 
The capacity of this software to easily interface with any kind of research organization guarantees that the research will be carried out effectively and achieve its goals.įacilitates fiscal administration by recording expenditures and receipts.įacilitates the beginning and maintenance of several studies at once. If you're looking for a clinical trial management system, Clinical Conductor CTMS is the best choice for you. The time and money needed to learn and implement this program are minimal. It allows for more organized budgeting by facilitating the creation and monitoring of invoices. There is a low threshold for supervision required to implement this software.Įnterprise resource planning is aided, which is useful for CROs (Contract research organizations). It can be used as a standalone system or as a plug-in to your current clinical trial management system to add functionality, connections, and interfaces that may be lacking. You may improve the effectiveness, efficiency, and manageability of your clinical trial operations with the help of BSI CTMS. Invoices and inventory data are now all kept electronically. The first is Quality Management System (QMS) Xpress, which is a ready-to-deploy solution for businesses currently digitizing their compliance process.Īdministration of quality control at suppliers With this system, you can choose from three different options that bring together quality management system (QMS) tools and clinical trial management (CTMS) processes. Includes methods for managing patients and finding new volunteersĭot Compliance is a clinical trial management system developed for the pharmaceutical, biotechnology, contract research organization (CRO), and medical device industries to guarantee documentation and compliance. EDGE's primary goals as a platform for collaboration are the elimination of duplication of effort and the safeguarding of patient data. This program's goal is to furnish scientists with a special and powerful environment in which they may monitor their investigations effectively from beginning to end and have instantaneous access to their data. White labeling/rebranding of mobile apps and websites for both mobile and desktop devicesĬonsistency with all wearables (smartwatches, fitness trackers, etc.) MainEDC ePRO enables patient access via mobile or web-based devices. MainEDCTM, powered by AI, streamlines even the most complex trials and studies for clinical research organizations (CROs), Big Pharma, and biotech. MainEDC is a clinical trial platform with a wide variety of integrated features, such as electronic data capture (EDC), an interactive web response system (IWRS), drug and supply management, and electronic patient-reported outcomes (ePro). You can contact the sales team to request a quote. In spite of the platform's intuitive design, encapsia offers the assistance of consultants who are trained to adapt it to the specific requirements of each client's research or business. With real-time data syncing, there are no longer any delays when checking up on patients or making adjustments to their home care plans.įurthermore, it facilitates the sharing of pertinent information amongst team members regardless of their physical location.Įncapsia's REST API provides the means for it to be integrated with other programs. Encapsia's versatility as a clinical trial management platform is further enhanced by its capacity to record clinical information both online and offline, independently of the method of data collection. This adaptable software product facilitates decentralized clinical trials by giving sites and organizations the freedom to construct their own hybrid protocols according to their own needs and any applicable regulations. In the list of the top tools, we have mentioned the Top 25 Clinical Trial Management Tools along with their features and pricing for you to choose from. In such a case, software designed specifically for managing clinical trials can help with this. Misinterpretation of data and carelessness could come from handling these tasks manually. Since the information obtained is essential to the patient's well-being, it is not prudent to leave anything to chance. Thousands of participants (patients, medical professionals, researchers, etc.), several clinical resource systems, and extensive data are all required for a successful clinical trial. 
More and more people are choosing to get treated, which has increased the complexity of clinical studies. 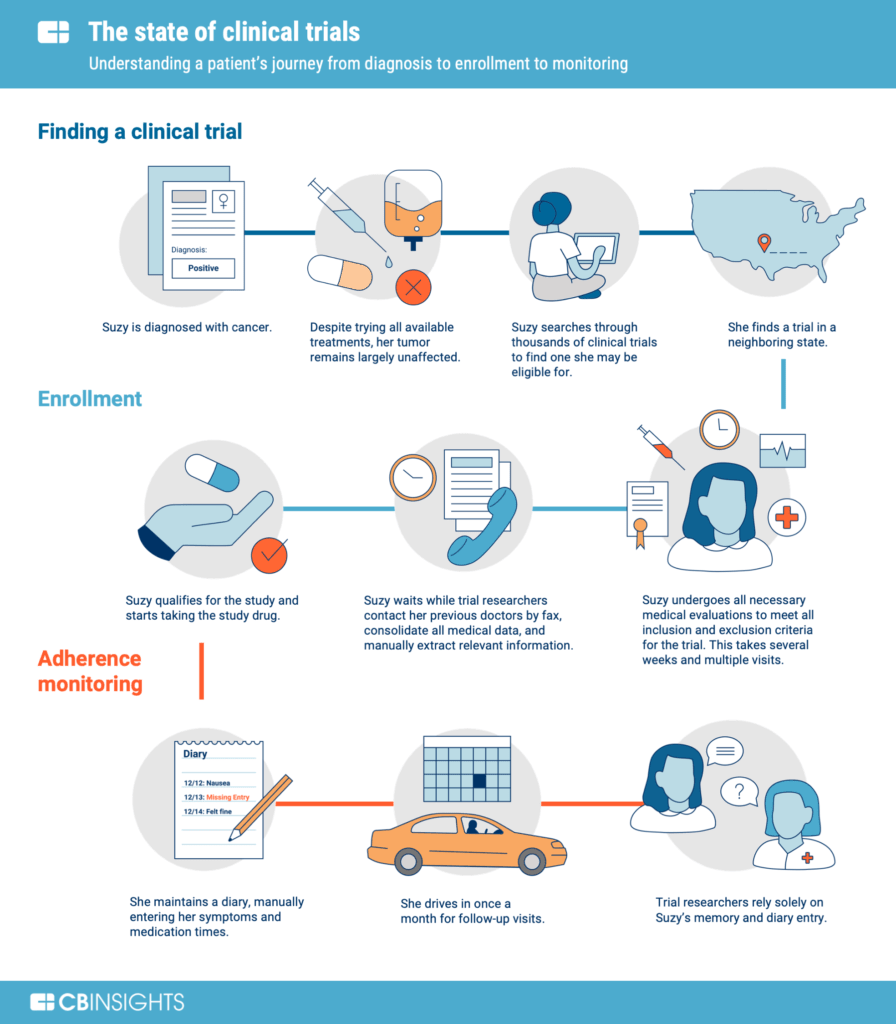 
Healthcare has been more accessible due to technological advancements, notably in the field of medicine. Clinical trial conduct is a massive and, at times, daunting undertaking. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
